Extending T1D Longevity: Balancing Lipids, Insulin, A1c
Prioritizing your health metrics leads to longer life
Ask any T1D what their A1c is, and most will probably know. Ask them what their LDL levels are, and most won’t know—or if they do, they won’t understand what it means. Or what to do about it. And that’s a big deal, as it can affect your lifespan a lot.
Let’s start with some startling facts that few T1Ds are aware of:
According to CDC Heart Disease Facts, 2024, cardiovascular disease causes about one in three deaths in the US. That is, a Major Adverse Cardiovascular Event (MACE). This is when plaque in your arteries (created by high LDL levels) break off and block blood flow, causing a heart attack or stroke.
For T1Ds, MACE is the leading cause of death, striking ~11 years earlier than in the non-diabetic population. More concerning is that some Americans die up to 15-17 sooner if they don’t have access to minimal healthcare.
Young adults with T1D under age 40 face a 20- to 40-fold higher risk of dying from heart disease compared to their peers.
This data puts to rest one of the most pervasive myths about T1D death: that you can die from hypoglycemia. The so-called “dead in bed syndrome”, as it was called, was thought the brain would die due to insufficient glucose. No autopsies have ever found that to be the case, except for severe and intentional overdoses of insulin by suicide, and even then, most attempts are unsuccessful.
For the vast majority of T1Ds, what severe hypoglycemia does is trigger acute and intense counterregulatory hormones to drive the liver to produce glucose. These raise blood pressure and cause other systemic responses that cause vulnerable plaques within the arteries to break off, triggering the heart attack or stroke.
It’s not the hypo that kills you, it’s the plaque in your arteries.
A note on younger T1Ds: Some may wonder how MACE explains “dead in bed” deaths in T1Ds under 40, who presumably haven’t developed significant atherosclerosis yet. Recent research clarifies this. A 2025 study in Frontiers in Endocrinology examined cardiac electrical changes during hypoglycemia in healthy T1Ds without cardiovascular disease. The authors explicitly state that pre-existing cardiac vulnerability, such as undiagnosed structural abnormalities or autonomic neuropathy in younger people, can result in the same outcome.
The saddest part is that the interventions that can avert heart disease are easier, cheaper, safer and in all other ways, simpler than struggling with the daily management of glucose levels. Better still, they can be done independently of glucose management.
That doesn’t mean glucose management isn’t important—it is. But it’s not the only thing T1Ds should focus on. And that’s what brings us back to those LDL levels that few T1Ds talk about or properly understand. Reduce the plaque, and you reduce your risk of death, potentially adding years to your life.
The aim of this article is to unpack all this to live a longer, healthier life. For that, let’s start at the end and work backwards.
The basics of cardiovascular disease
Cardiovascular disease (CVD) leads to a fatal heart attack or stroke—what doctors call Major Adverse Cardiovascular Events, or MACE. And it’s triggered by plaque that breaks off from artery walls, travels along until the path narrows too small to pass through, forms a blood clot, and, well, that’s it.
This is where LDLs come in. LDL stands for low density lipoproteins, which is colloquially called “bad cholesterol”. Once LDLs penetrate arterial walls, they trigger an immune response, causing macrophages to accumulate and form that plaque mentioned above. Over time, plaque builds up and becomes increasingly less stable. All you need is one big “event” that unlodges some plaque, and that’s it.
Let’s say your arteries are like the garden hose you connect to your water spigot. Plaque is like mud that’s stuck on the interior of the hose. If you turn the water spigot on, the amount of pressure might be strong enough that the shearing force can rupture the mud, where it then travels through until it gets to a narrow spot—like one of the hose kinks you often see—and then the mud gets stuck. Boom. Heart attack.
Short story: Eliminate the mud and there’s no risk of a heart attack. Put in terms of LDL levels: Clear all that out, and you reduce the plaque levels, thereby reducing your risk of death. By a LOT.
Glucose has nothing to do with this.
This is why I mentioned earlier that the intervention is relatively simple, cheap, and safe: a variety of medications are perfectly suited.
So, are we done here?
Not quite. It’s only 50% of the story.
It’s not feasible to reduce LDL levels to zero, so risk isn’t zero. But we can address the next thing in the cascade: Reduce the ability for those LDL particles to attach to artery walls in the first place. To do that, we need to bring glucose back into the conversation. Indirectly, at first.
Endothelium: The protective armor
The reason LDL can do this damage is that it penetrates a protective armor that lines the interior of your arteries: endothelium. It’s more like thin latex gloves protecting your hands. If endothelium is healthy, the LDL particles don’t break through. But if it’s damaged or destroyed, not only do LDLs get in, but the amount of damage correlates to the rate at which LDLs get in.
So, how do we keep the endothelium from getting destroyed?
That brings us to—not glucose—insulin. Yes, insulin.
You could spend your entire life priding yourself on achieving excellent A1c numbers—all the way down to non-diabetic levels—and still die prematurely from MACE, all because your LDL levels had resulted in plaque, which formed because all that insulin you took to keep those A1c levels lower eroded your endothelium.
This is not new; it’s been known since insulin was discovered. According to the article Insulin Resistance and Atherosclerosis : Common Roots for Two Common Diseases?, cardiologist Paul Dudley White noted in 1931 that “diabetes favors arteriosclerosis” and that an “excess of insulin” might contribute to this condition.
Arteriosclerosis is a disease of the arteries characterized by the deposition of plaques of fatty material on their inner walls. When you combine arteriosclerosis with cardiovascular disease, you get ASCVD—a term you’ll hear more often among cardiologists.
So, not glucose? No, not glucose. In the article, Diabetes Alters Activation and Repression of Pro- and Anti-Inflammatory Signaling Pathways in the Vasculature, the authors explain how excess insulin exposure—whether endogenous in T2D or exogenous in T1D—damages the endothelium through multiple converging mechanisms toward a chronically inflamed, pro-atherogenic state even in the absence of severe hyperglycemia.
To be clear here, insulin is not the only thing that affects endothelial function—it can also be damaged from smoking, hypertension (high blood pressure), chronic inflammation, and obesity/metabolic syndrome, the latter of which is also directly associated with excess insulin levels. But context is important.
Studies measuring endothelial function in children with T1D and studies involving smokers show that high insulin exposure causes similar degrees of vascular damage as smoking. People with Type 1 diabetes on insulin therapy show flow-mediated dilation values (a measure of endothelial health) comparable to those of active smokers—both groups showing roughly 50% impairment compared to healthy individuals.
We all know that smoking is bad. Not everyone is as aware that excess insulin levels are similarly harmful.
Let that sink in for a moment.
Another thing about excess insulin: it can trigger acute hypoglycemia. As described in the paper, Effects of Hypoglycemia on Cardiovascular Function in Patients with Diabetes, we learn that, during an acute episode, epinephrine and norepinephrine flood the bloodstream, dramatically increasing heart rate, cardiac contractility, and blood pressure.
An acute hypo in the middle of the night is often a cause of death for T1Ds. At one time, it was thought that death was due to the brain not getting enough glucose—a phenomenon called “dead in bed syndrome”.
But it wasn’t till decades later that autopsies didn’t find any evidence of brain damage, but lots of evidence of MACE. So, an acute hypo event in the middle of the night is like forcing a 55-year-old with high LDL levels to suddenly pop out of bed and sprint as fast as possible for 15-20 minutes. If there’s vulnerable mud (plaque), the mechanical stress from elevated blood pressure and heart rate can rupture the plaque, forming a clot that blocks the artery. Boom. Heart attack.
We T1Ds get acute hypos at night too often as it is. As you get into mid-life, if you have elevated LDL levels and a lifetime of elevated insulin levels, your risk profile is very, very high.
Who knew that insulin—the hormone that saves our life and keeps us from dying every day—is also responsible for a shortened lifespan.
Glucose: The Lethal Bullet
Up to this point, glucose didn’t really play a role. If you reduce LDL levels, your risk is reduced, and if you reduce insulin levels, you protect your endothelium, but naturally, you can’t reduce both to zero. Because of that, we now look at the role glucose plays. It does not initiate atherosclerosis, but it does amplify it.
The mechanism is glycation. Glucose binds irreversibly to proteins and lipids, forming advanced glycation end products (AGEs). The higher one’s glucose levels, the greater the number of AGEs in your body. Over time, this makes tissue less flexible and more brittle, affecting virtually every part of you.
When LDL proteins get glycated, the harms they’re already doing—forming plaque—are amplified. A lot.
You could have LDL levels of 80 mg/dL, where your doctor says, “we’ll keep an eye on it”, but if your A1c levels are elevated, each of those LDLs can do as much harm as though your levels were 120, 140, 200, or higher.
This is where A1c levels can be particularly damaging. People whose A1c levels are above 10% are at considerably higher risk of MACE if they have any appreciable levels of LDL.
(For the extensive detail, see the article, How hyperglycemia promotes atherosclerosis: molecular mechanisms and expanded in The Diabetes Mellitus–Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism.)
So, now we see all three come together: LDLs are the killers, excess insulin levels remove the protecting armor, and excess glucose levels assure that each bullet is more lethal than it would otherwise be.
The solution is to reduce the number of killers (reduce LDLs), keep the armor intact (reduce insulin), and make sure the bullets aren’t as lethal (reduce A1c levels).
One natural reaction to this is something that T1Ds have followed for years: low-carb diets. Let’s look at those.
Why Low-Carb Diets Don’t Work
Ironically, low-carb diets not only don’t help, they can actually make the problem worse, but from a different physiological angle: When carbohydrate intake drops too low, the body must mobilize massive amounts of stored fat to use as energy. To convert fat to glucose, the liver must package these free fatty acids into VLDL particles for transport, which eventually become the LDL that drives cardiovascular disease.
The amount of LDLs generated rises along with the amount of fat that needs to be converted. The lower the carb intake, the more fat is used. And the more energy you need—especially for exercise—the amount of fat utilization increases.
According to a meta-analysis in PLOS ONE, people with the lowest carbohydrate intake—such as the Bernstein diet—had the highest risk of overall mortality (32% increase), cardiovascular disease mortality (50% increase), and cerebrovascular mortality (51% increase).
But what about the low insulin levels? Shouldn’t that keep the endothelium intact?
Interestingly, insulin is biphasic in this way—it’s harmful at high levels and low levels. In the article Endothelial dysfunction in vascular complications of diabetes, researchers noted that insulin’s normal role is shown to be vasoprotective at physiological (normal) levels—but vasotoxic at both extremes: too high and too low. (“vaso” refers to “vasculature”.)
The key is avoiding extremes. Moderate carbohydrate intake (150-250g daily) and moderate insulin intake is the sweet spot. For a complete analysis of low-carb diets, see my article, The Paradox of Low-Carb Diets: A1c vs. Metabolic Health.
The sweet spot for ideal insulin levels—not too high and not too low—is 35-40 units of insulin per day, according to Dr. Ralph DeFronzo during this interview with Dr. Peter Attia. Dr. DeFronzo is one of the foremost experts in diabetes research, especially in the area of glucose and insulin research.
One’s total daily dose is one thing, but balancing between basal and food-based boluses is another challenge completely. For more, see my article, Basal Insulin Reduction: A How-To Guide using “Active Learning”.
Some may find it challenging to maintain healthy glucose levels with that amount of insulin per day, so let’s put glucose levels back in the spotlight.
Balancing Insulin, Glucose and LDL
To see how to balance insulin, glucose, and LDL, let’s look at those who live a long time with T1D. What are their profiles like?
Fortunately, we have lots of data to look at, beginning with the Golden Years Cohort, a group of 400 T1Ds in the UK who’ve had the disease for over 50 years. Their mean HbA1c was 7.6% (± 1.4), with some levels as high as 8.5%-9%. None had an HbA1c below 7%. And yet, they all had very low LDL levels, and modest total daily insulin of 0.52 u/kg/day.
Another group is the Joslin Medalists, American T1Ds who’ve also had the disease for over 50 years. They had an average A1c of 7.7%, and also had low lipid levels and insulin requirements (0.46-0.52 u/kg/day).
What they shared was metabolic health: normal weight, low insulin requirements, and crucially, low lipid levels. Neither group achieved this through perfect glucose control.
Ok wait — If glucose is so important, how is it that their A1c’s are so high and they lived so long?
They lived long because they had low LDL levels, but they also suffered a lot of microvascular complications due to their elevated glucose levels. In an analysis published in Diabetes Care, among the Joslin 50-Year Medalists with mean A1c of 7.7%:
57.4% had proliferative diabetic retinopathy (PDR)
13.1% had nephropathy (kidney disease), and
60.6% suffered from neuropathy.
Importantly, only 21.2% were free of all three microvascular complications. So, A1c levels do matter.
Let’s put these concepts together:
T1Ds who lived to 70+ were able to do so because they had low lipid levels.
However, decades of A1c levels > 7% resulted in eye, kidney and nerve diseases.
THEREFORE:
Lower lipids = live longer, and
Lower glucose levels = live better.
Most of my other articles deal with glucose management—exercise, diet, CGM use, insulin absorption variability, food absorption, and many other topics. I won’t cover those here. But what none of them cover is how to track and manage lipid levels, so now that we’ve covered how cardiovascular disease happens, let’s talk about lipid management.
Tracking and Evaluating Lipids
Virtually everyone who has any kind of diabetes should monitor lipid levels just as often as they get their A1c tests done. No exceptions, and no age limit. Yes, children too.
Lipids should be included in all blood work orders, and should include a ‘lipid panel’ that includes total cholesterol, LDL cholesterol, HDL cholesterol, and triglycerides. These are the minimum, but you should also include apoB and Lp(a), if those aren’t included in that default panel.
The particles that erode your endothelium are LDL, VLDL, IDL, and Lp(a), but they all share one structural feature: they each contain exactly one apolipoprotein B (apoB), and it’s this protein marker that indicates atherosclerotic risk. This is why apoB is the most important one to test for.
Lp(a) is entirely genetic, so you only need to test for it once. If it’s elevated, you need to be hyper-aggressive in treatment (next section).
If you don’t get apoB, knowing your LDL levels is the next best thing, but note that people with insulin resistance or elevated triglycerides may appear to have acceptable LDL levels, but their particle count (apoB) could be dangerously high. This discordance explains why some people with “normal” LDL still experience cardiovascular events.
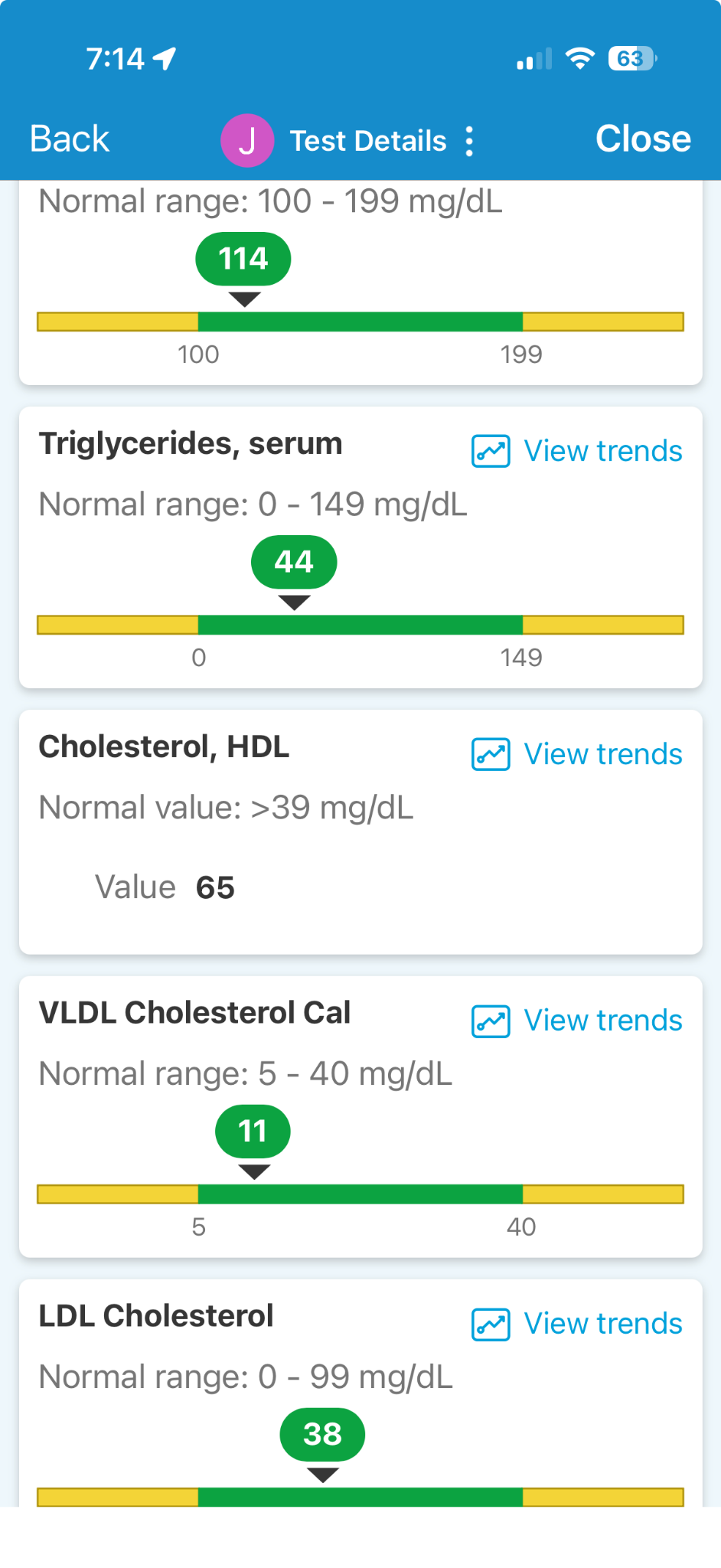
When you get results, labs report the values along a line that also indicates the “normal” range. Here’s an illustration, with the first item being “total cholesterol”.
In this lab report, everything is in the green “normal” zone, but this should not be confused with “safe”. In this case, the LDL level is 38, which is unusually low, but this person is taking medications to reduce their LDL levels because they are aware that risk rises with higher levels.
Without this medication, levels would be considerably higher—even as high as 80—at which point, doctors will say, “we’ll keep an eye on it.”
Unfortunately, few clinicians are as up to date on cardiovascular risk as they are on the latest insulin pumps, so when they see “normal ranges” for lipids, they’re not thinking about T1Ds vs. non-diabetics. So, they typically don’t act. And this is one of the leading reasons why T1Ds die from MACE so early in life: Because elevated insulin levels can erode endothelium, even “normal” levels of LDL can be harmful because the protection is weaker, and doctors aren’t prescribing medications to lower LDL levels.
And remember, there are also AGEs from elevated glucose levels, which make those same LDLs even more lethal, far more than that of the general population. By the time you’re in your mid-50s, you may have had “normal” LDL levels, but your compromised endothelium and weaponized LDLs dramatically elevates your risk. An acute hypoglycemic event could be the big one that dislodges vulnerable plaque.
So, your response to the doctor is, “No, you’re not going to keep an eye on it. You’re going to lower it.”
That leads to the next step: interventions.
Lipid Management
There is no healthy or safe level of LDL, apoB or Lp(a). The fewer harmful particles there are, the less able they are to create the “vulnerable plaque” that leads to MACE. You want to go as low as medically possible.
An analysis in PMC emphasized the dose-response relationship: the lower the LDL, the lower the risk, with no apparent threshold. This isn’t just correlation—it’s mechanistic confirmation. Fewer atherogenic particles mean less substrate for plaque formation.
So, how should you reduce LDL levels?
Doctors and media say that lipid levels can be lowered through diet, exercise and pharmacology. This is technically true…for the general population, where risk profiles are much lower. But, T1Ds’ risk of MACE is so substantial, that the goal is not to compare yourself against the general population. It’s impossible to eat low fat food and exercise a lot and hope to reduce LDL and apoB. Yes, those are good habits to have, and there are other benefits, particularly for diabetics. But they are rarely going to lower harmful lipid levels on their own. You will need pharmacological interventions (drugs).
Fortunately, the drugs to do this are old, well-established, well-tolerated, inexpensive, and generally available through most medical insurance.
Statins are the foundation of lipid therapy, blocking cholesterol synthesis in the liver and typically reducing LDL by 30-50%. They’re well-tolerated, inexpensive ($4-20/month generic), and have decades of safety data. Common options include atorvastatin and rosuvastatin. About 5% of the population can experience muscle soreness to varying degrees with statins.
A detail that comes up is that statins can induce insulin resistance, but it manifests more dramatically in non-diabetics because it degrades beta cell function. Most who aren’t at risk of T2D do not see a concern. It’s only when people start gaining weight, whether including both non-diabetics and diabetics (T1 and T2) that insulin resistance is a problem. The real insulin resistance is due to visceral fat, at which point, statins can amplify the existing resistance. That progression eventually leads towards T2D. Many studies see this correlation and some even attribute it to “causality”, which is a stretch.
As for T1Ds, the beta cell function is not an issue of course, but then we get into T1D obesity, which is now “double diabetes” territory, and we have the same situation as noted above: Statins will increase the resistance proportionally to the degree of obesity. Studies show T1D insulin dosing increasing by up to 10% in these cases. While that’s not good, it’s also only 10%, a modest increase that’s nearly irrelevant in the grand scheme of things: Weight gain is the real problem.
Now assemble the risks: Statins are reducing MACE by huge proportions, while potentially contributing to some added insulin resistance due to conditions that are already present and should be addressed anyway (weight loss).
Ezetimibe (Zetia) works differently—it blocks cholesterol absorption in the intestine, adding another 15-20% LDL reduction when combined with statins. It has minimal side effects and costs roughly $10-30/month generic. Note that many studies show Ezetimibe has minimal effect when used alone—you need to take it with either statins or PCSK9 inhibitors.
PCSK9 inhibitors (evolocumab/Repatha, alirocumab/Praluent) are the most powerful option, reducing LDL by an additional 50-60% on top of statins by increasing the liver’s ability to clear LDL particles from the bloodstream. Because they don’t have the confounding effects of statins, they’re becoming very popular in this domain. They are currently taken by injection every other week, but an oral version is in trials and expected to be released in coming years.
While expensive ($500-600/month without insurance), many insurance plans now cover PCSK9 inhibitors for high-risk patients—yes, T1Ds—and patient assistance programs exist.
The results speak for themselves: The FOURIER trial, published in the New England Journal of Medicine, followed 27,564 patients with established cardiovascular disease. Those treated with evolocumab (a PCSK9 inhibitor) achieved median LDL levels of 30 mg/dL—far below what was thought safe or beneficial. The result: a 15% reduction in major cardiovascular events and, critically, continued benefit down to LDL levels as low as 10 mg/dL with no safety signal.
The recently published VESALIUS-CV trial in NEJM went further, studying patients without prior heart attacks or strokes (primary prevention). Among 13,970 participants, evolocumab produced a 25% reduction in major cardiovascular events and a stunning 36% reduction in myocardial infarction. The benefit was consistent across all subgroups, including those with diabetes.
A 2024 review in Drugs on long-term PCSK9 therapy documented both excellent tolerability (minimal side effects even after years of treatment) and evidence of plaque modification on imaging. The substrate itself becomes less dangerous.
Children and Lipid Medications
All the medications listed above are FDA approved and appropriate for children, typically starting around age 8-10. Statins have 20 years of safety data showing no effects on growth, development, or long-term health. PCSK9 inhibitors and ezetimibe also have strong pediatric safety profiles. The key is working with a pediatric lipidologist who understands appropriate dosing and monitoring.
A 20-year statin study showed that starting treatment in childhood prevented early cardiovascular death compared to starting in adulthood. Zero cardiovascular deaths in the children who started statins early. At age 39: 100% survival.
The cumulative LDL burden matters - every year of elevated LDL damages arteries. Starting early = more years protected.
Summary
The conventional wisdom in T1D care has been almost entirely on glucose control—chasing perfect A1c numbers. And yet, cardiovascular disease has silently claimed lives decades too early.
The big takeaway is that you can win at both: Medications to lower apoB levels lead to a longer life. Managing glucose levels leads to a healthier one.
The Joslin Medalists survived to 70+ without perfect glucose control—but they also paid the price with eye, kidney, and nerve damage. You don’t have to accept that trade-off. With modern CGM technology and the knowledge in this article, you can optimize both.
Your next steps are simple:
At your next appointment, request a complete lipid panel including apoB and Lp(a).
If elevated, advocate for treatment - the medications are safe, proven, and inexpensive.
Continue managing your glucose, but now with the confidence that you’re addressing the real killer.
Building intelligent glucose-management habits is 20% technique and 80% psychology. For more, see my article, The Four Habits of Healthy T1Ds.
As for me, you can read how I managed through all this in my article, Why I Haven’t Died Yet: My Fifty Years with Diabetes.



Neat stuff, Dan. I recently started my own blog on this topic (Diablog) and I enjoy reading your substack. I’m
a cancer researcher who developed LADA type diabetes, so I’m applying what I know about signaling, stem cells, and the immune system to diabetes. Your substack has been quite informative.
Thanks so much for this awesome article. I think you might have persuaded me to explore statins (t1 for 32 yrs, in good health and low a1c, but keep being offered them and instinctively saying no!).